Thư viện tài liệu, ebook tổng hợp lớn nhất Việt Nam
Website chia sẻ tài liệu, ebook tham khảo cho các bạn học sinh, sinh viên

 Bài giảng Sinh thái vi sinh vật - Chương 3: Tương tác giữa các vi sinh vật
Bài giảng Sinh thái vi sinh vật - Chương 3: Tương tác giữa các vi sinh vậtGiới thiệu chung - Cùng với sự phát triển của các quần thể vi sinh vật thì nhu cầu về dinh dưỡng và không gian sống của vi sinh vật cũng ngày càng tăng lên. Điều này khiến cho các vi sinh vật biến đổi không ngừng để có thể tồn tại được trong môi trường sống. - Vi sinh vật không chỉ phản ứng với môi trường hóa học mà chúng còn tương tác với các vi...
 21 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 26 | Lượt tải: 0
21 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 26 | Lượt tải: 0 Bài giảng Sinh thái vi sinh vật - Chương 2: Môi trường sống tự nhiên của vi sinh vật
Bài giảng Sinh thái vi sinh vật - Chương 2: Môi trường sống tự nhiên của vi sinh vậtGiới thiệu chung - Bên trong hệ sinh thái có rất nhiều khu vực mà vi sinh vật cư trú, được giới hạn bởi các điều kiện vật lý, hóa học và sinh học được gọi chung là nơi sống (habitat) - Vi sinh vực chiếm giữ và thích ứng với ổ (niche) bên trong nơi sống giống như ở động vật và thực vật. Tuy nhiên khả năng thích ứng của chúng đối với các chức năng ...
 13 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 25 | Lượt tải: 0
13 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 25 | Lượt tải: 0 Bài giảng Sinh thái vi sinh vật - Chương 1: Tổng quan về sinh thái vi sinh vật và sự đa dạng của vi sinh vật
Bài giảng Sinh thái vi sinh vật - Chương 1: Tổng quan về sinh thái vi sinh vật và sự đa dạng của vi sinh vậtGiới thiệu chung Sinh thái học vi sinh vật là gì? Microbial ecology = Microbial + Ecology Sinh thái học vi sinh vật nghiên cứu mối tương tác giữa vi sinh vật với môi trường của chúng bao gồm môi trường vật lý, hóa học và sinh học. Nghiên cứu về sinh thái học nhằm mục đích: - Xác định thành phần loài vi sinh vật hiện có - Vai trò của mỗi loài ...
 17 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 31 | Lượt tải: 0
17 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 31 | Lượt tải: 0 Tối ưu hóa quá trình thủy phân chế phẩm protein từ rong Cheatomorpha sp. bằng enzyme Alcalase
Tối ưu hóa quá trình thủy phân chế phẩm protein từ rong Cheatomorpha sp. bằng enzyme AlcalaseTrong nghiên cứu này, ảnh hưởng của các yếu tố như: pH, nồng độ enzyme và thời gian thủy phân đến quá trình thủy phân protein rongCheatomorpha sp. được nghiên cứu. Phương pháp bề mặt đáp ứng được sử dụng để tối ưu hóa điều kiện thủy phân nhằm thu được peptide có hoạt tính sinh học cao nhất. Kết quả cho thấy, điều kiện thủy phân tối ưu để thu được d...
 9 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 26 | Lượt tải: 0
9 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 26 | Lượt tải: 0 Bài giảng Nhập môn Sinh học phân tử - Nguyễn Thanh Tố Nhi
Bài giảng Nhập môn Sinh học phân tử - Nguyễn Thanh Tố NhiLược sử phát triển Gồm 3 giai đoạn: - Giai đoạn tiền đề - Giai đoạn sinh học phân tử ra đời - Giai đoạn sinh học phân tử hiện đại
 39 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 28 | Lượt tải: 0
39 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 28 | Lượt tải: 0 Bài giảng Sinh học phân tử - Đột biến gen
Bài giảng Sinh học phân tử - Đột biến genKhái niệm - Đột biến = các biến đổi di truyền xảy ra đột ngột. Về bản chất, đột biến gen là một sự thay đổi trong trình tự nucleotid trong bộ gen. - Tác động của thay đổi trình tự gen: - Không có tác động: đột biến lặn → đa hình về trình tự - Tạo ra một protein đột biến với trình tự acid amin thay đổi - Tác động đến quá trình điều hòa ...
 61 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 35 | Lượt tải: 0
61 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 35 | Lượt tải: 0 Bài giảng Sinh học phân tử - Điều hòa hoạt động gen
Bài giảng Sinh học phân tử - Điều hòa hoạt động genĐIỀU HÒA HOẠT ĐỘNG GEN - Điều hòa lượng sản phẩm của gen được tạo ra = gen có được phiên mã và dịch mã hay không - Điều hòa hoạt động gen ở prokaryote: chủ yếu xảy ra trong quá trình phiên mã - ĐIỀU HÒA PHIÊN MÃ (KIỂM SOÁT PHIÊN MÃ) + KIỂM SOÁT CẢM ỨNG ÂM + KIỂM SOÁT ỨC CHẾ ÂM + KIỂM SOÁT CẢM ỨNG DƯƠNG - ĐIỀU HÒA HẬU DỊCH MÃ (KIỂM SOÁT SAU D...
 32 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 26 | Lượt tải: 0
32 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 26 | Lượt tải: 0 Bài giảng Sinh học phân tử - Sinh tổng hợp protein
Bài giảng Sinh học phân tử - Sinh tổng hợp proteinKHÁI QUÁT - Sinh tổng hợp protein = Dịch mã = Giai đoạn cuối của sự biểu hiện TTDT/mARN thành trình tự acid amin tƣơng ứng/chuỗi polypeptid - Xảy ra trong tế bào chất - Ở Prokaryote khác eukaryote (eukaryote phức tạp hơn)
 52 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 31 | Lượt tải: 0
52 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 31 | Lượt tải: 0 Bài giảng Sinh học phân tử - Các loại ARN
Bài giảng Sinh học phân tử - Các loại ARNARN - Là phân tử trung gian trong quá trình biểu hiện gen - Sản phẩm của phiên mã - Được tổng hợp trong nhân (eukaryote), vào TBC tham gia tổng hợp Protein
 55 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 24 | Lượt tải: 0
55 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 24 | Lượt tải: 0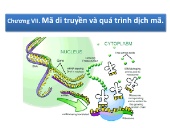 Bài giảng Sinh học phân tử - Chương VII: Mã di truyền và quá trình dịch mã
Bài giảng Sinh học phân tử - Chương VII: Mã di truyền và quá trình dịch mã*Mã di truyền Mã bộ 3: cứ 3 Nu trên phân tử ADN mang thông tin qui định trình tự axit amin trên phân tử polypeptit. Tính chất: • Tính phổ biến : mã di truyền giống nhau hoặc rất gần giống nhau ở tất các các loài sinh vật. Có một số trường hợp ngoại lệ xảy ra ở ty thể người, nấm men và một số loài khác, ở đó mã UGA quy định tryptophane khác so vớ...
 17 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 31 | Lượt tải: 0
17 trang | Chia sẻ: Tiểu Khải Minh | Ngày: 17/02/2024 | Lượt xem: 31 | Lượt tải: 0

